3TR Researchers Develop Core Outcome Measures Sets for Children and Adults With Severe Asthma (COMSA)
Led by the 3TR consortium, a European multi-stakeholder working group has developed 'Core Outcome Measures sets for paediatric and adult Severe Asthma' (COMSA). Core outcome measures (COM) are a set of tools that should be used in all studies about a specific topic. Read more about the COMSA study in this recent publication of the 68-member group of authors and watch the accompanying video specially produced by the 3TR working group and the communications team. To better understand the concept of COM, take a look at this short video. The COMSA will serve as a basis for future clinical trials and allow researchers to compare results between studies more easily. They were formed under the leadership of Ekaterina Khaleva, Graham Roberts, Ratko Djukanovic and Anna Rattu from the University of Southampton, and developed by representatives from across Europe and North America.
Until now, asthma researchers have not used a standard set of COM to understand whether or not biological therapies work. The international working group has filled this gap with the COMSA, which will enable better data creation and assessment of biologics in childhood and adult asthma clinical trials. The study was supported by the European Lung Foundation (ELF) and European Federation of Allergy and Airways Diseases Patients' Associations (EFA) to understand the perspectives of people living with severe asthma. With this strong backing, the following COM were selected by the 3TR researchers, patients, clinicians, pharmaceutical representatives, and health authorities.

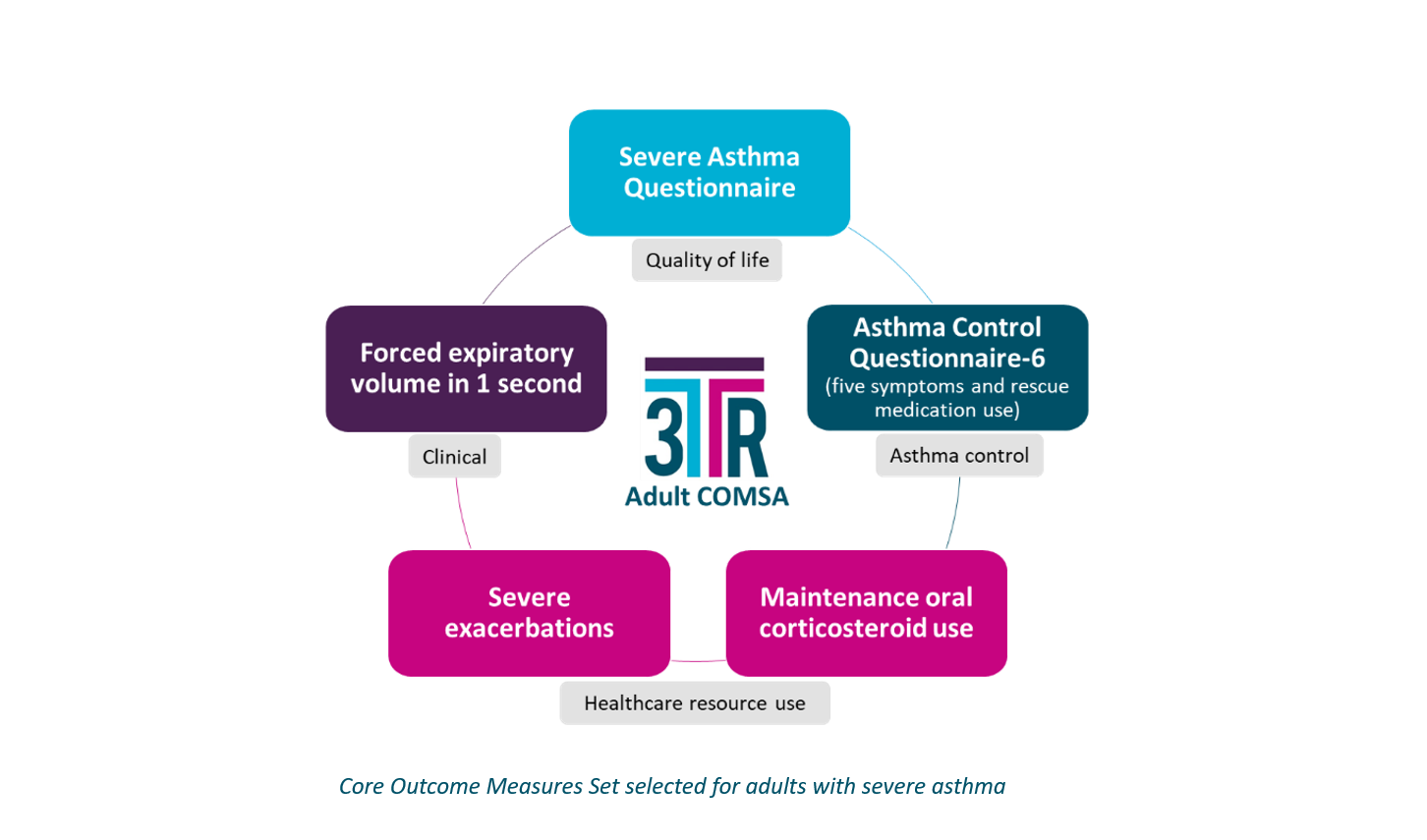
3TR's asthma experts expect that the results of this patient-centred collaboration will:
- Help inform which COM are used in future clinical trials
- Improve the ability to compare how well biologic medicines work
- Help assess the value for money of biologic medicines
- Improve therapy for people with severe asthma and maximize their quality of life
Below, COMSA participants reflect on their experiences of this project and how the COMSA will change the future of severe asthma research and care:
Helen Parks, 3TR Respiratory Patient Working Group representative:
I found the experience to be incredibly insightful and felt that I was contributing to a meaningful activity. The patient voice is key in my mind to ensuring a good outcome for all parties. There is nothing more demoralising than feeling you are ignored as the patient. Contributing in this process was rewarding, enjoyable and made me feel I could help other patients with severe asthma have a better experience than I have had as a patient. Sometimes how asthma can impact on day-to-day life is more important to the patient than say a medical aspect. In this way COMSA gets the widest perspective and encourages patient input which is vital.
- Does your doctor know about the COMSA? Share this new tool with your healthcare team so your care is personalized and considers core outcome measures that we know are important to patients.
Ekaterina Khaleva, paediatric clinician, researcher:
During the developmental process we especially wanted to ensure that COMSA include outcomes that are important to each stakeholder group. What is particularly beneficial is that COMSA will harmonize the way outcome measures are reported in clinical studies and enable comparisons of data to produce meaningful results. I think working together with other stakeholder representatives is essential to succeed in the development of novel approaches to patient-centred management and improving long-term outcomes of patients with severe asthma.
- Evaluating the success of a biological treatment for your patients living with severe asthma? Consider using the paediatric and adult COMSA to improve patient outcomes.
Vera Mahler, clinician, health authorities’ representative:
From the health regulator’s perspective, the development of core outcome measures sets for paediatric and adult severe asthma (COMSA) is of great value. Until now clinical trials on the treatment of severe asthma have lacked standardized outcome measures, hampering the comparability of efficacy and effectiveness of different therapies. Using the newly developed COMSA in future trials can help to overcome these problems and ideally will lead to improved reporting, assessment and comparability of trial results. This concerns not only the regulatory tasks but also serves the understanding of the treating physicians and finally enables a more patient-orientated treatment decision making.
Graham Clarke, clinician, pharmaceutical representative:
At AstraZeneca, we are committed to developing novel therapies that can change the lives of patients suffering from severe asthma. The development of patient-centred outcome measures is a valuable tool with the ambition to inform the design of future clinical trials and better assess the impact of novel therapies emerging from our pipeline.
- Are you planning a clinical trial to evaluate the effectiveness of biological treatments for severe asthma? Consider using the COMSA as your core outcome set for more comprehensive and patient-centred results, that can be easily compared across studies.